

|
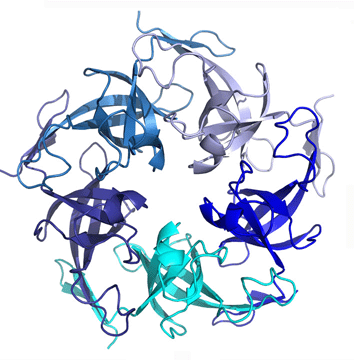
The Yeates laboratory is located at UCLA in the Department of Chemistry and Biochemistry. Our research covers the areas of molecular, structural and computational biology. In structural biology, our emphasis is on supramolecular protein assemblies, such as self-assembling protein filaments, layers, and cages. Supramolecular assemblies of interest include both natural and designed structures. Much of our current focus is on bacterial microcompartments, which are giant virus-like structures that serve as primitive organelles inside many bacterial cells. We have recently determined the three-dimensional atomic structures of several proteins that make up the shell of the carboxysome, a microcompartment that performs CO2 fixation, providing the first detailed clues about how it operates. Strategies are also being developed for designing novel proteins to assemble into ordered structures on the mid-nanometer scale. In the areas of computational biology and bioinformatics, our recent studies have led to the discovery of unusual microbes that are able to stabilize their proteins at extreme temperatures through mechanisms such as widespread disulfide bonding, and the use of topological complexity like linking and knotting. We also continue to work on developing new methods for deciphering protein function and protein interactions by applying ideas in symbolic logic to genomic data. |  |